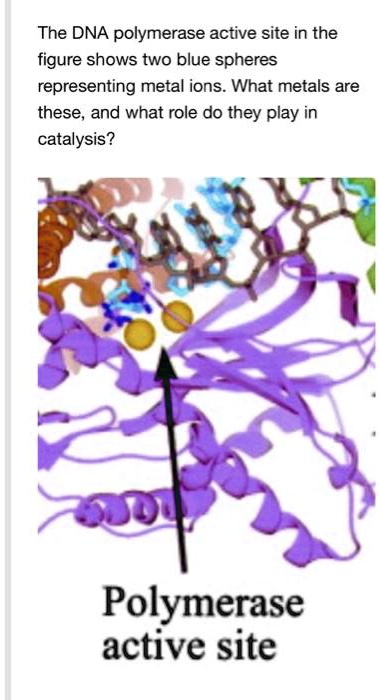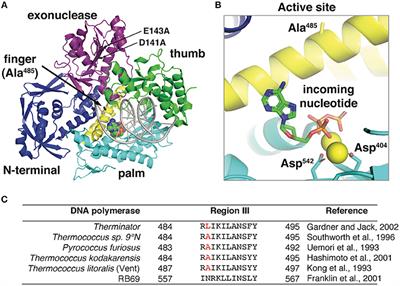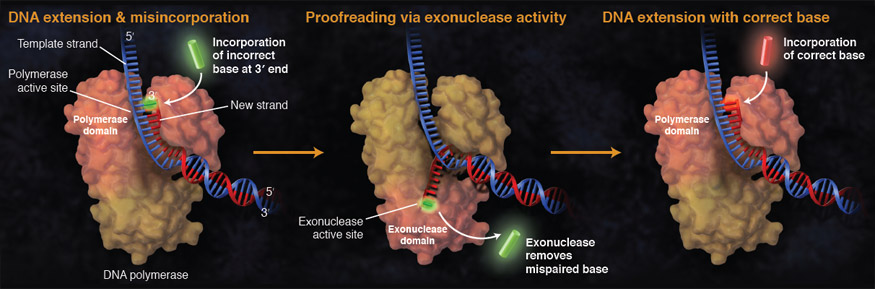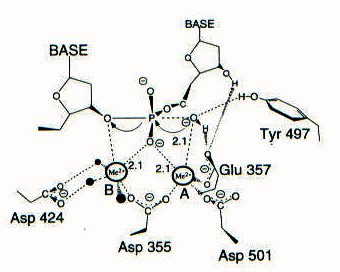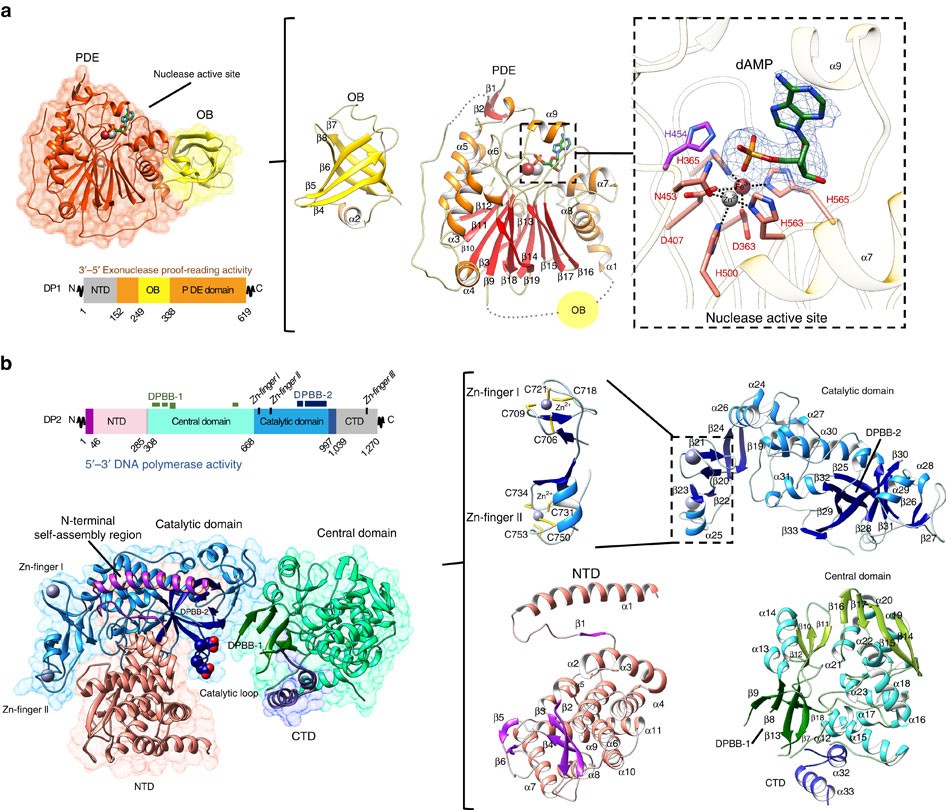
Shared active site architecture between archaeal PolD and multi-subunit RNA polymerases revealed by X-ray crystallography | Nature Communications

The Structural Basis for Processing of Unnatural Base Pairs by DNA Polymerases - Marx - 2020 - Chemistry – A European Journal - Wiley Online Library

Insight into the mechanism of DNA synthesis by human terminal deoxynucleotidyltransferase | Life Science Alliance

Highly Organized but Pliant Active Site of DNA Polymerase β: Compensatory Mechanisms in Mutant Enzymes Revealed by Dynamics Simulations and Energy Analyses: Biophysical Journal

Figure 4 from Crystal Structure of the Catalytic α Subunit of E. coli Replicative DNA Polymerase III | Semantic Scholar

Critical Role of Magnesium Ions in DNA Polymerase β's Closing and Active Site Assembly | Journal of the American Chemical Society
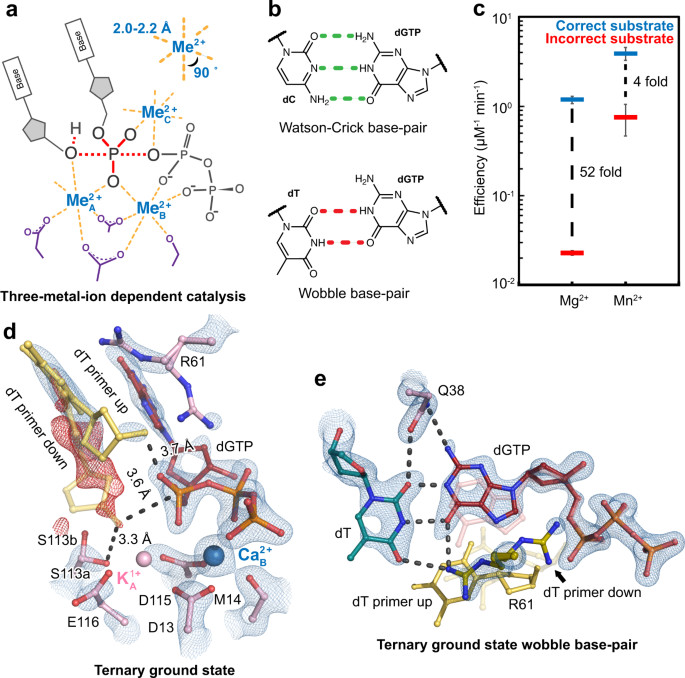
In crystallo observation of three metal ion promoted DNA polymerase misincorporation | Nature Communications
A Model for Transition of 5′-Nuclease Domain of DNA Polymerase I from Inert to Active Modes | PLOS ONE
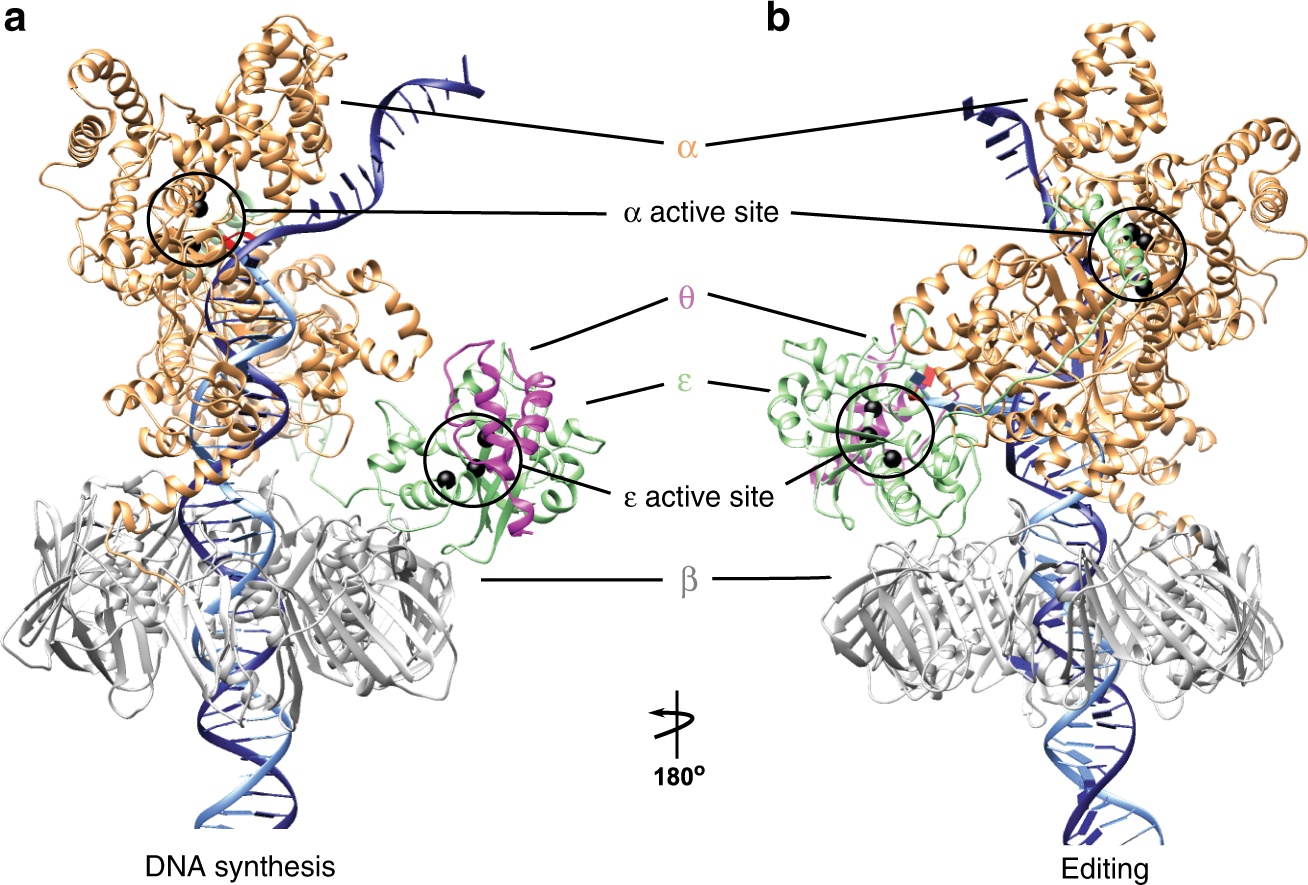
Polymerization and editing modes of a high-fidelity DNA polymerase are linked by a well-defined path | Nature Communications
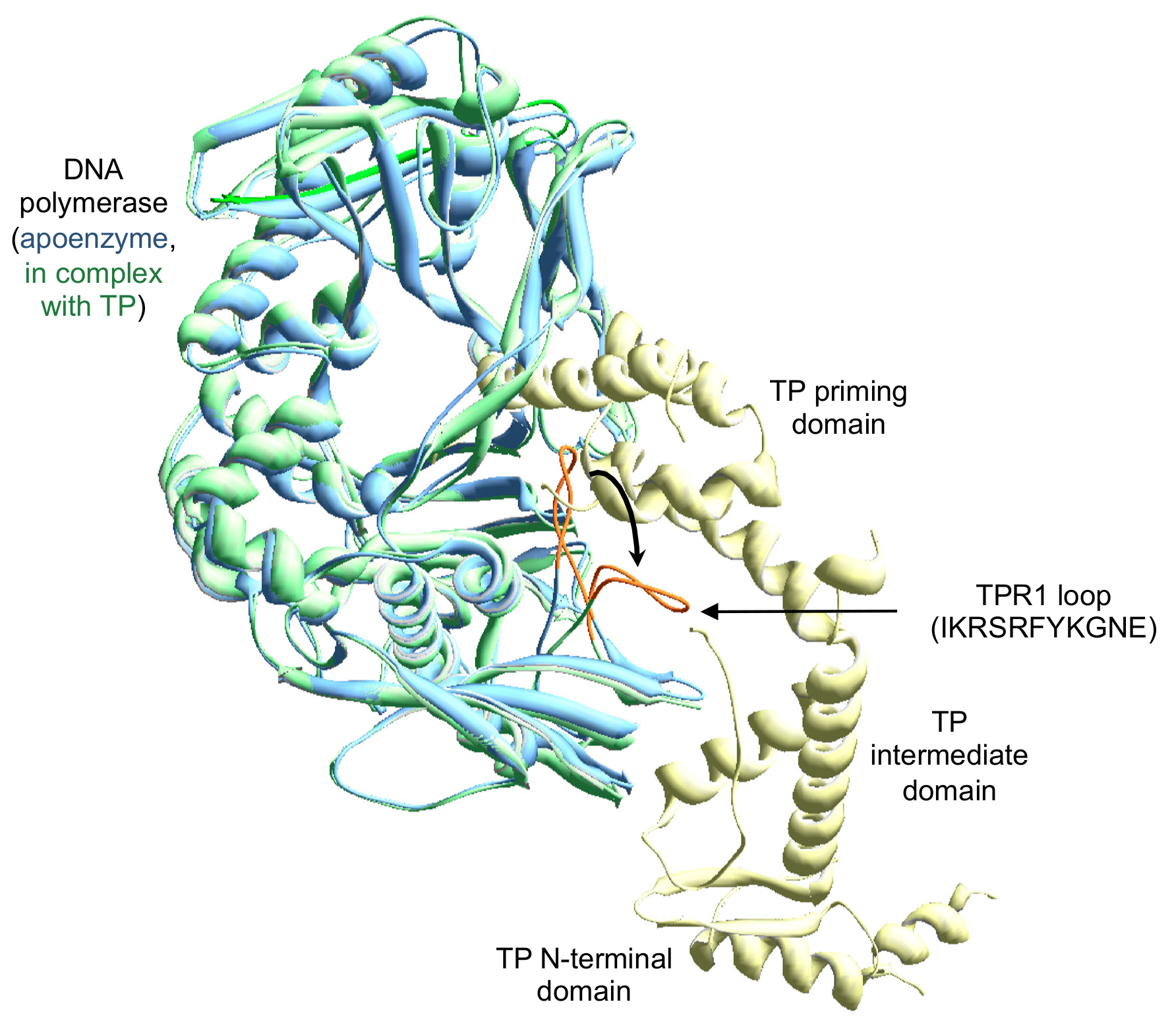
Biomolecules | Free Full-Text | The Loop of the TPR1 Subdomain of Phi29 DNA Polymerase Plays a Pivotal Role in Primer-Terminus Stabilization at the Polymerization Active Site

Critical Role of Magnesium Ions in DNA Polymerase β's Closing and Active Site Assembly | Journal of the American Chemical Society